Contact A Stryker Knee Replacement Attorney
Based on the information that has been found since the Class 1 FDA recall, it seems Stryker was aware or should have been aware of many of its medical device problems earlier. However, the company failed to investigate or analyze adverse event reports involving the Strkyer Triathlon knee replacement device or to perform proper design validation. There were other Stryker knee replacement devices with major problems as well.
If you have been injured by a defective Strkyer knee replacement device, you may be entitled to compensation for your pain and suffering, medical costs, lost earnings, and loss of enjoyment of life.
Our attorneys will determine if you are eligible to join a class-action lawsuit, keeping in mind that such legal matters take time. It could be easier in your case to secure an individual settlement faster.
Please contact our law office today for a free legal case review.
References
Take Action If Youre Hurt By A Defective Knee Replacement Device
You do not have a lawsuit simply because the knee replacement device that was implanted in your body has been recalled. However, if the device fails prematurely and you suffer a physical injury or require additional surgery, then you may have a claim against the pharmaceutical company that designed, made, or marketed the knee replacement device.
Our Massachusetts knee replacement lawyers are here to help you if youve been hurt. Contact us today for a free, no-obligation consultation about your rights and about how we may be able to help you get the full and fair recovery that you deserve.
|
Related Links: |
Tips For People Who Are Filing Lawsuits
Some tips if you are considering filing a lawsuit:
- Dont delay! The law limits the time you have to file a lawsuit, and the deadline varies from state to state. Speak with an attorney as soon as possible so you can file before the statute of limitations closes.
- Consult an attorney before participating in a manufacturer recall. Some recalls require you to waive your right to participate in a lawsuit, which would prevent you from receiving compensation.
- If you have not yet had your revision surgery, make arrangements with an attorney to have the explanted device and tissue samples properly stored as evidence.
- Gather all your medical and pharmaceutical records in one place.
Recommended Reading: How To Lighten Your Knees Overnight
Cemented Total Knee Replacement
After the patients are medically cleared for surgery and received appropriate anesthesia, the knee joint is opened from the front. The diseased segments of the bones forming the joint are removed. Using cutting guides, bone cuts made in the femur, tibia and the patella.
Cemented TKA
After thoroughly washing the cut segments, acrylic bone cement is applied to the implants and the bone segments. The implants are then fixed to the prepared bed on the bone.
A plastic high-quality polyethylene space is attached to the tibial component to reduce friction between the implants. The acrylic cement hardens quickly providing immediate fixation.
Constrained polyethylene insert
A constrained polyethylene insert may be used in a knee with ligament instability on the sides. The tall and wide tibial post on the polyethylene insert ensures rotational and small side to side stability. The insert is made of highly cross-linked polyethylene which is resistant to wear and tear. The tibial post fits in front of the femoral cam providing stability.
Femoral Component
The posterior stabilized femoral component involves sacrificing the posterior cruciate ligament during surgery. The function of the posterior cruciate ligament is duplicated by the femoral cam and the tibial post mechanism.
Modular femoral component
Modular femoral stem
Modular femoral component
Modular tibial component with bone augments
Complete Summary Of Knee Replacement Recalls 2017

Register to receive a free US Orthopedic Large Joint Device Market Report Suite synopsis and brochure
The year 2017 saw a substantial number of knee replacement recalls by the FDA, all of which were classified as Class 2 device recalls. The FDA defines a Class 2 recall as a situation in which use of, or exposure to, a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote. An FDA recall order forces the manufacturer in question to take action towards addressing a problem with a certain device in violation of FDA law.
In addition to FDA forced recalls, medical device manufacturers may issue voluntary recalls in which a potential problem might risk their reputation. The list of knee replacement recalls for 2017 below consists of only FDA recalls.
Zimmer Biomet
Most notably, on February 22nd, Zimmer Biomet was issued a total of 5 recall orders pertaining to their Oxford product line due to potential alumina inclusion in the finished product that could lead to cracking and separation. Products affected included the Oxford Partial Knee System Right and Left Medial Tibial Tray , Femoral Slap Hammer and Tibial Resector Body Tube & Guides, as well as the Oxford Unicompartmental Knee Phase 3 Tibial Impactor and various polyethylene implants: Sports Med, Repicci II Tibial Components.
Other knee replacement recalls by Zimmer Biomet in 2017 included:
| Date |
Smith & Nephew
Also Check: How Do I Get Rid Of Fat Around My Knees
Recent Advances In Knee Surgery
Since the first modern knee replacement surgery was performed in 1968, surgeons and researchers have made many attempts to improve the techniques, materials, and technology that are involved to achieve greater success.
Some recent innovations in knee replacements include:
- Minimally invasive surgery: this allows for smaller incisions and faster healing. However, it also requires great skill and precision on the part of the surgeon and some have argued that lesser technicians risk poorer outcomes.
- Cement-less knees: in traditional knee surgery, cement is used to bond the knee implants with the existing bones. In some cases, the cement can break down, causing devise failure. In cement-less knees, the bone naturally fuses with the implant. However, some studies suggest that cement-less knees can pose device failure problems of their own.
- Mobile-bearing vs. fixed-bearing implants: some companies offer a plastic insert that is able to rotate and offer greater movement for a patient compared to a fixed-bearing implant, in which the plastic spacer is firmly connected to the metal component.
- Posterior cruciate ligament-retaining vs. -substituting implants: in which the patients ligament is used rather than replaced to create greater stability.
- Gender-specific designs: tailored to the female anatomy for greater functionality. There has been some debate as to whether these gender-specific designs are effective.
Complications And Risks Of Knee Replacement Surgery
Most patients who choose to have knee replacement surgery are very happy with their . Typical patients find relief of pain, and an ability to increase their activities. However, there are possible complications of the surgery, and why knee replacement patients may not be happy. Here are five problems that can frustrate patients who have knee replacement.
Also Check: Can I Regrow Cartilage In My Knee
Common Reasons For Knee Replacement Recalls
Medical device manufacturers issue recalls of knee replacement implants for many reasons. Some recalls may be due to simple matters of improper packaging, but others may involve defective components that can cause serious injuries. Common recall issues for knee replacement joints are:
- Poor design: An improper design may cause fractures in tools or implants. Some tools have a flawed design that can cause them to fall into the surgical site.
- Improper fit: An implant can be damaged if a surgeon forces the device into place in the hip joint.
- Loosening: Some replacement joints can loosen over time and require revision.
- Early wear: Metal or plastic parts may wear much faster than expected.
- Co-mingled components: Parts of the right and left knee may be mixed and cause surgical complications.
Zimmer Knee Replacement Litigation
There are currently some 1,000 Zimmer knee replacement lawsuits pending in the courts. These product liability lawsuits claim failure to warn, negligence and design defect as the primary offenses. Plaintiffs argue defective and dangerous NexGen knee components are the result of a failure to perform sufficient pre-market safety testing.
Qualifying federal NexGen knee lawsuits have been consolidated into a multidistrict litigation before Judge Rebecca R. Pallmeyer of the U.S. District Court of the Northern District of Illinois. Bellwether cases are set to begin in early 2015.
Recommended Reading: How To Stop Limping After Knee Surgery
Meet Our Knee Replacement Attorneys
You dont have to suffer alone. Let us fight for fair compensation for your medical bills and suffering.
Typically used for less-active patients. Range of improvement is 20-50 percent.
Mobile-Bearing Prosthesis:Allow for slightly more rotation than fixed-bearing prosthesis. Soft tissues must be strong enough to support this option, or knee dislocation is possible. These implants typically last longer and allow for more active lifestyles, but the surgery is more challenging.
Posterior Cruciate Ligament-Retaining:The PCL is the ligament that runs along the back of your knee. These implants allow the doctor to keep your PCL intact, giving you a greater range of motion.
Posterior Stabilized:If this implant is used, the PCL is removed, and the implant serves as the stabilization for the back of your leg, keeping your thigh bone from going too far forward when you bend your knee.
Bicruciate-Retaining Designs:These implants are built to allow the patient to retain both the posterior cruciate ligament and the anterior cruciate ligament, allowing for the best range of motion and flexibility.
Gender-specific:Knee implants were once unisex, but manufacturers have developed components that are thought to fit better with the average womans knee. However, your surgeon should be choosing your implant based on your unique anatomy.
Problems With The Nexgen Cr
Zimmer sought to alleviate the problems with existing knee replacements that used cement to secure the implant to the bone. This cement could leech into the patients bloodstream and enter the soft tissue surrounding the knee, leading to inflammation and more serious complications. Zimmers solution was the use of a cobalt-chromium alloy and porous fiber to fuse the implant to the existing bone.
Typical knee replacements have a life expectancy of 10 to 15 years, but according to a non-peer reviewed report presented by former Zimmer consultant Dr. Berger to the American Association of Orthopedic Surgeon- the CR-Flex appeared to fail in as little as 2 years. Over one-third of the 108 participants he observed suffered from radiographic loosening, and 8.3% required revision surgery.
Zimmer has consistently maintained that the failure rate is indicative of improper surgical techniques and not because of inherent issues with the design of the product itself.
Complications associated with the NexGen implants include:
- Component loosening
- Soft tissue damage
- Nerve damage
You May Like: How To Reduce Swelling After Knee Replacement
Are Aesculap Knee Replacement Devices Safe
Patients who have undergone knee replacement surgery and received Aesculap knee replacement devices are reporting painful side effects. Many have been forced to undergo follow-up procedures to repair or remove their devices.
The FDA has received reports about an increasing number of incidents since the beginning of 2016 regarding the need for revision surgery for recipients of Aesculap Knee Replacement systems.
Patients are reporting problems with the loosening of their devices.
In many instances, surgeons performing the revision procedures have noted a complete absence of the bonding cement in follow-up surgeries. One surgeon performed 16 revision surgeries on devices that were no more than four years old and found all of the devices had been affected by cement debonding.
In many cases, revision surgeries require surgeons to use a saw to remove the device because the adhesion between the bone and the replacement device is so strong.
Surgeons have reported being able to remove Aesculap components without any assistance, simply pulling components from the patients body by hand because there was no bond between the device and the bone.
Knee Replacement Revision Surgery
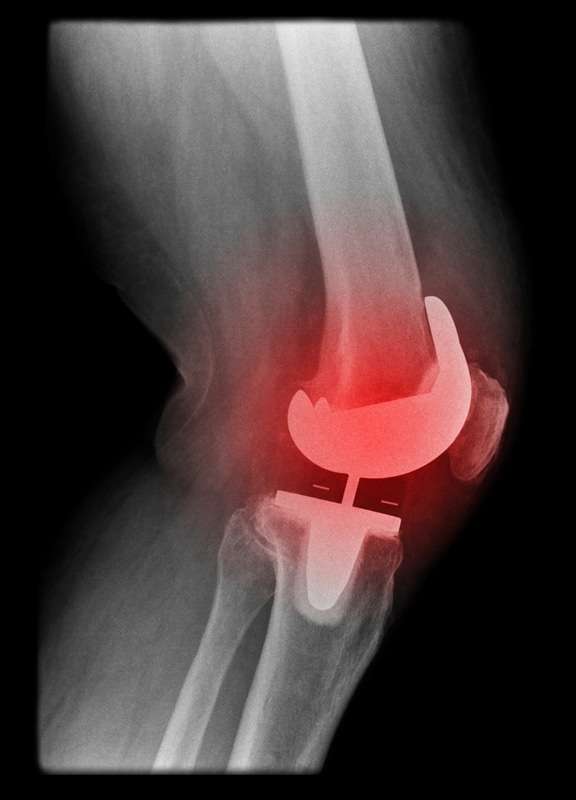
Revision surgeries are expensive. The average cost of revision surgery is nearly $50,000.
Revisions surgeries are also more likely to result in extended hospitalization. The average hospital stay for a knee revision is slightly more than 5 days. Revisions also require follow-up care and physical therapy.
Don’t Miss: How To Get Rid Of Cellulite Above My Knees
Zimmer Knee Replacement Systems
Zimmer has been one of the most trusted names in knee replacement for over four decades. Started in 1968, Zimmer, Inc. took the lions share of the knee replacement market in 1995 with the release of the FDA approved NexGen Complete Knee Solution. Since being brought to the market, almost 500,000 knee replacements have used this particular artificial knee.
In 2001, NexGen Flex components were introduced to give the knee greater overall movement. This advance was meant to give patients the ability to engage in a much more active lifestyle than the previous version. The flex components were also gender specific and advertised as fitting patients better and being just as reliable as the previous components.
The FDA cleared the new components under the 510 process, where the Complete Knee Solution qualified as significantly equivalent to a device already on the market, and therefore didnt require clinical trials. The 510 fast-trackclearance process is an issue of contention among physicians because the absence of clinical trials relieves the manufacturer of the burden of proof of safety. This led to Zimmer being able to market products that might ultimately injure those they are meant to help.
NexGen knee replacement systems by Zimmer:
What Should You Expect After Knee Revision Surgery
Most patients who undergo revision procedures can expect to have good to excellent results. Although expected outcomes include pain relief with increased stability and function, complete pain relief and restoration of function is not always possible.
Up to 20% of patients may still experience some pain following revision knee surgery. This can persist for several years after the procedure. Additionally recovery after revision total knee surgery is heavily dependent on the state and function of the knee prior to the revision surgery.
You May Like: Getting Rid Of Fat Around Knees
Zimmer Persona Knee Replacement Lawsuit
The Food and Drug Administration approved the Zimmer Personalized Knee System in late 2012. Just three short years later, Zimmer implemented a product recall for the Trabecular Metal Tibial Plate, a component of the system, because of reports that it may cause problems requiring revision surgery.
Zimmer announced the recall in February 2015. About 11,658 devices are affected. In addition, the company also recalled the Persona Tibial Articular Surface Provisional Shim, a tool used during knee replacement surgery, because of the potential for problems.
Patients who went through a knee replacement with the Zimmer Persona Knee and later experienced pain, loosening, and other problems are urged to contact a personal injury attorney. Revision surgery can be complicated and often requires a longer recovery than what is typical after an initial replacement surgery. Families may be entitled to compensation to help cover medical expenses.
What Is A Knee Replacement
The knee is made up of a complicated set of tendons, muscles, ligaments, and bones. When the joints become damaged by either progressive arthritis, trauma, or other destructive diseases it can cause severe pain that limits everyday activities or even moderate to severe pain while resting during the day or night. In these circumstances, knee replacement surgery can help restore quality of life.
According to the American Academy of Orthopedics, more than 700,000 total knee replacement surgeries are performed each year in the United States, and nearly 90% of patients who undergo knee replacement report a dramatic reduction of knee pain following surgery. Knee implants allow for normal activities and motion and typically last 15 to 20 years or more.
There are more than a dozen manufacturers of artificial knee implants. A variety of issues can cause knee replacement failures.
Read Also: How To Whiten Knees And Elbows
How Did Knee Implants Get Approved By The Fda
The first knee replacement implants were proposed in the 1880s, when a German surgeon suggested that ivory implants held in place with cement could replace damaged knee joints.
Modern knee replacement research picked up in the 1950s, with the development of replacement joints using metallic components. In the 1970s, researchers first debut knee replacements that allowed for the preservation of the posterior and anterior cruciate ligaments.
Recent innovations in knee replacement implants have focused on allowing for greater range of motion, so that younger patients can stay more active.
The experienced attorneys at McIntyre Law are now handling knee replacement lawsuits. Let us help you.
Long Term Knee Replacement Problems
Longer term knee replacement problems are rare but include:
1)Wearing of the Prosthesis: 85% of knee replacements last at least 20 years2)Loosening of the Prosthesis: 1.4% of patients require a repeat total knee replacement, known as a revision knee replacement3)Infection in the Knee Joint: this can occur years after surgery
Don’t Miss: Is Nano Knee Covered By Medicare
What Makes Someone Require A Knee Replacement
Most people who receive knee replacement surgery have arthritis. But injury, other diseases, or simply years of use can also damage the knee.
Your doctor may recommend knee replacement surgery if:
- Your knee pain prevents you from sleeping.
- Your knee pain prevents you from doing everyday activities such as walking.
- Your knee pain prevents you from taking care of yourself.
- Other treatments have failed to reduce your knee pain.
Depuy Revision Rates And Recalls
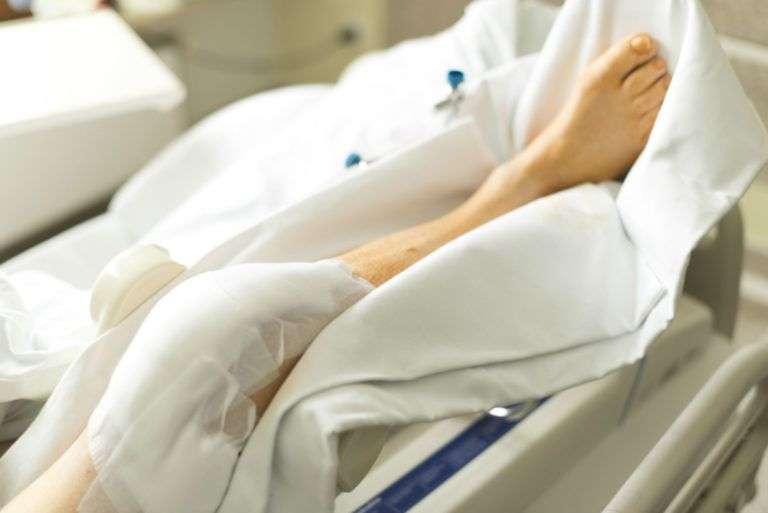
Other recent issues with DePuy knee systems have led to increased rates of revision surgeries. Revision surgery is done when an artificial knee component or system fails and needs to be replaced. This may occur if the knee system has reached the end of its expected life, with normal wear and tear causing it to break down, but revisions are also needed when knee systems fail prematurely or causes damage or pain.
Several DePuy knee systems have been found in recent years to cause higher numbers of revision rates than are expected. The high revision rates indicate that something is wrong with the system or a component, and can lead to a recall. In 2015 several thousand LCS Complete knee system inserts were recalled because of exceptionally high revision rates in Australia.
In the U.S. in 2017 higher revision rates were being reported for a Sigma implant. Called the Sigma HP PFJ cemented trochlear implant, this component was implicated in the high number of revisions in patients who had received the knee system. The implant had the potential to cause the joint to come out of alignment, to become unstable, or to become dislocated, all issues that can lead to the need for revision surgery. DePuy issued the safety warning, recalled existing parts, and then discontinued the implant.
Read Also: Why Does My Knee Hurt When It’s Cold