What Zimmer Persona Lawsuits Are There
The Zimmer Persona Knee recall affected nearly 12,000 components that may have been implanted in patients who were placed at risk of early device failure. Many people have filed lawsuits to seek compensation for injuries caused by their Zimmer Persona knee device.
Zimmer-Biomet is not new to recalls or lawsuits. Over the past several years, the combined company has faced multiple recalls and thousands of lawsuits for harm caused by Zimmer and Biomet devices such as the Durom Cup, a hip device, and its NexGen knee devices. Some Zimmer-Biomet lawsuits have been settled but many may remain in various courts.
Several of these lawsuits have been successful. Zimmer Biomet has paid out $56 million in lawsuit settlements just in 2012 alone. Zimmer settled the remaining active lawsuits regarding its knee replacement devices in 2018.
Notwithstanding claims relating to this product, the drug/medical device remains approved by the U.S. FDA.
What Metal Is Used In A Zimmer Knee Replacement
In Group 1 , the surgeon used a fiber metal femoral component and an uncemented Trabecular MetalTM monoblock tibial component, both of which were approved by the FDA.
Certain Trabecular Metal knee implant components have been identified as a hazard, according to Zimmer Biomet. In the packaging, there is a chance of tears and holes. When this occurs during surgery and is not detected, the patient may be implanted with a non-sterile implant. A non-sterile implant is only as likely to cause an infection as it is to cause one. If you or someone you care for recently had knee replacement surgery, and if you have any questions or concerns about this procedure, please contact your surgeon or the hospital.
Djo Enhances Portfolio With Acquisitions And Partnerships
DJO has said that it is building toward a $1 billion joint replacement business through new partnerships and technology.
DJO previously invested in strategic innovations, including those from Insight Medical Systems, developer of ARVIS . ARVIS combines tracking cameras with a 3D display and hands-free interface in an integrated eyepiece for total joint replacement DJOs EMPOWR Partial, Primary and Complex Primary Knee and Hip systems are compatible with ARVIS.
After acquiring Mathys, a Switzerland-based joint replacement company, in June 2021, DJO expanded its joint replacement channels outside the U.S. The deal has the potential to nearly double DJOs international revenue, with an expanded global market for the AltiVate Reverse shoulder replacement, EMPOWR Hip and Knee Systems and foot and ankle products. Among the products that Mathys brings to the partnership is the RM Pressfit vitamys Monobloc acetabular cup.
PeekMed and DJO Surgical recently developed a new technological strategic partnership at the end of 2021. The partnership leverages the respective strengths of DJOs surgical business and PeekMeds dedicated focus and expertise as an innovative technological player for orthopedic surgeons.
Recommended Reading: What’s Good For Your Knees
A History Of Product Issues
About a month before Zimmer recalled its Persona knee replacement components, it sent an urgent notice to doctors, hospitals, and surgeons, telling them to immediately quarantine all Persona components. Its a process that Zimmer is all too familiar with.
In recent years, many medical devices have been pulled from the market for failure rates, and as one of the largest device manufacturers in the world, Zimmer has been among the companies required to do so.
In 2008, Zimmer instituted a short recall for its Durom Cup hip replacements. Zimmer representatives claimed that the recall was necessary because surgeons performing the procedure were not trained well enough. However, several experts, including one of Zimmers own paid consultants, found that the implants circular edge made it nearly impossible to keep it in place.
In 2010, Zimmer was required to recall approximately 70,000 MIS Tibial devices, also due to early loosening. And in 2014, the companys flagship product, the NexGen Knee replacement, couldnt escape the pattern of recalls when more than 40,000 were removed due to defective screws.
There have been several waves of Zimmer knee lawsuits as a result of its defective implants, and they have partitioned $70 million alone for damages resulting from the Durom Cup. Similar funds have been allocated for NexGen and MIS failures, and other funds will likely be set aside due to Persona failures.
Zimmer Biomet Announces One
One-Year Results Reinforce Interim Findings: mymobility Remote Care Management Platform on Apple Watch and iPhone Yielded Similar Outcomes as Traditional Care and Resulted in Reduced Outpatient PT and Surgery-Related ER Visits After Knee Replacement
WARSAW, Ind., Nov. 5, 2022 /PRNewswire/ — Zimmer Biomet Holdings, Inc. , a global medical technology leader, today announced one-year data from a multicenter, prospective, randomized controlled trial to evaluate the impact of mymobility® with Apple Watch, a first-of-its-kind remote care management platform. The data show that using mymobility with Apple Watch following primary knee arthroplasty, commonly known as knee replacement, can effectively guide rehabilitation, demonstrate similar outcomes to traditional care models, and significantly reduce the number of outpatient physical therapy visits. In addition, use of mymobility with Apple Watch was associated with significantly fewer surgery-related emergency department visits, which could translate to lower costs of care*. The data will be presented at a podium session at the 2022 annual meeting of the American Association of Hip and Knee Surgeons .
Key data findings include:
With 90+ years of trusted leadership and proven expertise, Zimmer Biomet is positioned to deliver the highest quality solutions to patients and providers. Our legacy continues to come to life today through our progressive culture of evolution and innovation.
|
Media |
Also Check: How Do I Treat Knee Pain
How Zimmer Biomet Is Approaching Remote Patient Monitoring
The pandemic forced the entire healthcare industry and its stakeholders into a more remote environment. Although the industry was slowly moving toward remote patient monitoring, COVID-19 accelerated the trend by years.
“We were forced to go remote with everything that we were doing, and so people innovated quickly, and looked at what potentially they could do to be able to reach patients and make sure they were getting the best care possible even if they couldn’t come into the office,” Liane Teplitsky, president of global robotics and technology & data solutions at Zimmer Biomet, told MD+DI in a September 2021 interview. “I know we did a pretty quick turn on our mymobility platform to make sure that it was just more easily accessible to any surgeon out there.”
Zimmer Biomet partnered with Apple a few years back to develop the mymobility digital care management platform that uses iPhone and Apple Watch to help orthopedic surgeons deliver support and guidance to patients through a more connected experience. Mymobility is designed to deliver continuous data and patient-reported feedback to facilitate care, outcomes, and patient satisfaction about surgical preparation and recovery. So, when COVID-19 hit, the company made sure its physician customers had that patient engagement component to deliver pre-op care, and even post-op physical therapy remotely.
A second major driver of the remote patient monitoring trend is that consumers are becoming increasingly tech savvy.
Does A Knee Replacement Add Weight
Weight gain is usually a side effect of joint replacement surgery. In a 2010 study conducted by the University of Delaware, 207 adults had knee replacements and gained an average of 14 pounds in the two years following the operation.
According to a 2010 study, the average person gained 14 pounds after knee replacement surgery. Patients who have knee replacement have a higher chance of losing weight than those who have hip replacement. The way you were before your knee replacement does not mean that you will be the same person after it. You have options to make your calories work for you and not against you. Eat on smaller plates and bowls when you see a meal that looks small on a larger plate, it indicates that it is the right size. The results can be significant if you allow them to work that way.
There is also the possibility that the surgery causes an increase in fluid volume. A number of factors, including the use of painkillers and surgery stress, can cause fluid retention after surgery. In the case of weight loss surgery, the body may be unable to expel all of the extra water, resulting in postoperative edema. In addition to weight gain, postoperative edema can cause bloating, fatigue, and low blood pressure, all of which are side effects. If you are concerned about your weight after surgery, consult with a doctor. A physician can help you lose weight while also keeping you healthy.
Don’t Miss: How To Save Your Knees
Zimmer Biomet Focuses On Data Capture
Much of Zimmer Biomets knee and hip replacement efforts have focused on components for ZBEdge, the companys suite of connected digital and robotic technologies, including ROSA, mymobility and the new Persona IQ knee.
In 2021, Zimmer Biomet gained FDA 510 clearance of the ROSA Hip System for robotically-assisted anterior total hip replacement, adding to the companys ROSA Robotics portfolio that includes the ROSA Knee System for total and partial knee replacement. FDA also granted Zimmer Biomet and Canary Medical a De Novo classification to market their Persona IQ, reportedly the first and only smart knee cleared by FDA for total knee replacement. Persona IQ combines Zimmer Biomets Persona The Personalized Knee with Canary Medicals implantable canturio te tibial extension sensor technology that measures and determines a patients range of motion, step count, walking speed and other gait metrics.
Data from these ZBEdge technologies are incorporated into Zimmer Biomets intelligence platform, OrthoIntel, and then provided to surgeons to inform patient care decisions.
What You Need To Know
- Product liability lawsuit filed against medical device supplier Zimmer Biomet alleging defective knee replacement instrument.
- Biomet recalled its Regenerex Series A Patella knee replacement device in 2017.
- Plaintiff’s four-count claim alleges product liability, failure to warn and breaches of express and implied warranty.
A recalled knee replacement system has kicked off new product liability litigation in Georgia after a recipient alleged portions of the medical device sheared inside his knee.
And its not the first time the medical device company has been accused of producing defective instruments.
Recommended Reading: Outside Knee Pain When Bending
What Difference Does The Odep Rating Make To Patients
Its fair to say I very rarely have a conversation with a patient who has chosen Yorkshire Knee Clinic because we use the PPK implant . It is, however, quite common for patients to ask which implant we will be using.
> Discover more about knee replacements
Often, they will have done their research and they will know that the partial knee replacement implants used most frequently are the PPK or ZUK, which are fixed bearing implants, and the Oxford Partial Knee, which uses a mobile bearing.
Yorkshire Knee Clinic favours and uses fixed-bearing implants which we believe offer both excellent functional outcomes and low risk of implant failure and in our opinion the implant most likely to achieve the best of these is the Persona Partial Knee.
In my opinion, the PPK is achieving the best implant survival data of any knee so far with excellent outcome scores in a range of studies, including outcome data gathered during our own use of the implant which we have presented to the European Knee Society.
For patients, thats good news, because it means they can place a lot of confidence in their knee, feeling secure in the knowledge that it will usually not only help resolve the pain they have been experiencing, but also that it is a real solution for the long term.
Zimmer Biomet’s Smart Knee: A Surgeon’s Perspective
Amanda Pedersen | Jul 06, 2022
BTIG medtech analysts recently hosted a fireside chat with Jeff Yergler, MD, an orthopedic surgeon who specializes in joint reconstructive surgery and is based in South Bend, IN. According to a report from BTIG’s Ryan Zimmerman, Yergler is among the earliest adopters of Persona IQ, Zimmer Biomet’s recently-launched “smart knee” implant, with more than 200 procedures already under his belt.
“We wanted to get Dr. Yergler’s perspective on the knee implant itself, the procedural considerations, the benefits of the data the implant generates, and how Dr. Yergler sees smart implants growing in the future,” Zimmerman wrote. “We came away from the call with the view that inside the operating room, Persona IQ, is just another knee implant . There are no trade-offs clinically, no extra time needed in surgical technique, and it pairs well with Rosa … Outside the OR there are still some early logistical hurdles to work through in order to make Persona IQ seamless between broader patient ecosystem .”
Zimmerman added that he expects Persona IQ to become more harmonious with each software update and tibial stem/sensor iteration. Perhaps more importantly, the analyst said the longer-term potential of “smart knees” and other implants is significant. He noted that Canary Medical, Zimmer Biomet’s partner on Persona IQ, is working to generate data that can provide predictive analytics in order to boost clinical outcomes.
Read Also: Why Do My Knees And Ankles Hurt
What Is Zimmer Knee Replacement Made Of
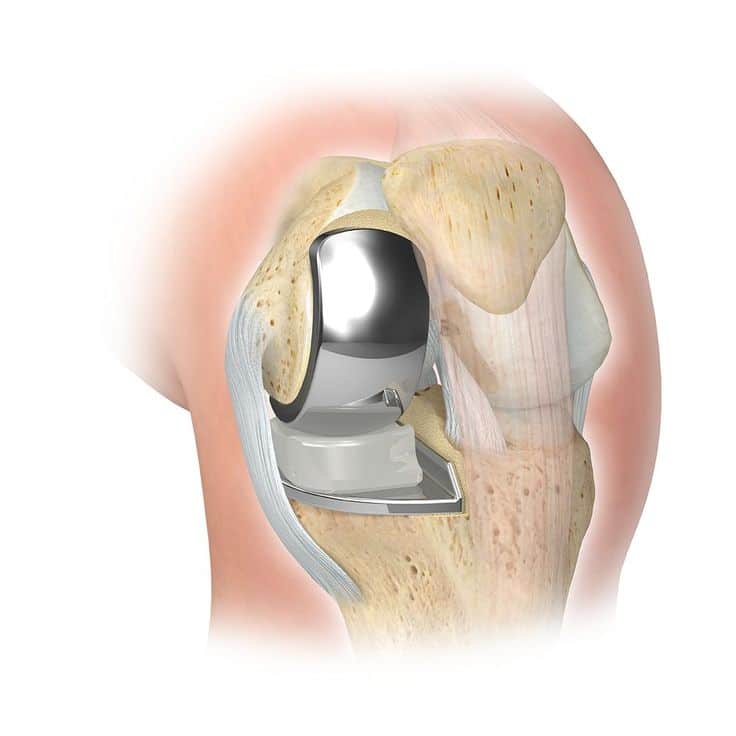
Zimmer knee replacement is a type of surgery that is used to treat knee joint problems. The knee joint is a weight-bearing joint that is made up of the femur , tibia , and patella . The ends of these bones are covered with a smooth, slippery material called articular cartilage. This cartilage allows the bones to glide over each other and absorb shock. Over time, the cartilage can break down, causing the bones to rub against each other. This can lead to pain, stiffness, and swelling. In some cases, the damage is so severe that the knee joint no longer functions properly. When this happens, knee replacement surgery may be recommended. During knee replacement surgery, the damaged cartilage and bone are removed and replaced with artificial components. These components may be made from metal, plastic, ceramic, or a combination of these materials. Zimmer knee replacement is one of the most common types of knee replacement surgery.
Zimmer, a global leader in the design, development, manufacturing, and marketing of reconstructive orthopaedic implants and fracture management products, is based in Warsaw, Indiana.
The Zimmer-Biomet Persona® Total Knee Cruciate-Retaining Femoral Component , which is used in conjunction with the kinematic alignment surgical technique, has been shown to improve functional outcomes and patient satisfaction following total knee arthroplasty.
Zimmer implants are made in the United States of America with the finest titanium metal.
Which Company Is Best For Knee Replacement

Zimmer is the most popular manufacturer of knee replacement implants. In fact, according to Zimmer, more than 25% of all knee replacements globally are manufactured by the company.
The Risks And Benefits Of Knee Replacement Surgery
People with severe arthritis should consider knee replacement as an option. Despite this, there are some risks associated with knee replacement, so it is critical to weigh those concerns against the benefits before making a decision. A potential problem with placement of the implant during knee replacement surgery is one of its risks. The joint instability can cause chronic pain, as well as revision surgery. There is also the risk of infection during knee replacement surgery. The risk of infection is highest for knee replacement surgery. Infection can spread to nearby tissues and cause death if not treated promptly. Before making a decision about knee replacement surgery, you must consider the risks and benefits of the procedure. You can get less invasive treatments in addition to the more traditional options, but you should look for the one that is best for you.
Recommended Reading: My Knees Hurt When I Walk
Who Needs A Total Knee Replacement
When a persons knee joint becomes severely damaged due to disease or injury, he or she may be a candidate for total knee replacement surgery.
Chronic, severe knee pain can be caused by many factors, the most common of which is arthritis. Arthritis is characterized by inflammation and stiffness in the joints and can reduce a persons range of motion. Arthritis can be caused by a disease or a traumatic injury.
Most knee pain is caused by three types of arthritis, according to the American Academy of Orthopaedic Surgeons , including osteoarthritis, rheumatoid arthritis and post-traumatic arthritis.
Zimmer Biomet: Vanguard System Will Revolutionize Total Knee Replacement
By Jof Enriquez, Follow me on Twitter @jofenriq
Orthopaedic products manufacturer Zimmer Biomet has unveiled Vanguard Individualized Design , which is touted as a first-of-its-kind total knee arthroplasty personalized construct, with dual bearings to better preserve the natural tissues surrounding the knee joint.
Most traditional knee implants feature a one-piece bearing made of polyethylene placed between metal portions of the implant. With Vanguard ID, Zimmer Biomet furthers this common design, incorporating two individual polyethylene bearings, with different articulations, on the medial and lateral sides of the knee. Orthopaedic surgeons can choose the exact specifications thickness and geometry of each bearing depending on the patient knee’s unique anatomical features and the surgeon’s specific preferences.
“I believe that our newly released Vanguard Individualized Design will revolutionize total knee replacement. With the ability to fine tune the knee’s balance through the use of independent medial and lateral bearing thickness and constraint options, we will arm the surgeon with the ultimate soft tissue respecting capability,” said Todd Davis, VP and GM of Zimmer Biomets global Knee business, in a news release.
Also Check: How To Make Your Knees Stop Hurting After Running
Beyond The Byline: Smart Knee Implant Tests Uncharted Territory
Zimmer Biomet added a sensor from company Canary Medical to a knee implant to create its “smart” implant.
Modern Healthcare Senior Hospital Operations Reporter Alex Kacik and Technology Reporter Jessica Kim Cohen discuss the latest tech development in joint replacement surgeries.
Music Credit: Coffee by Cambo
Related Article
Alex Kacik: Hello, and thanks for joining us. Modern Healthcare’s Beyond the Byline here, where we offer a behind-the-scenes look into our recording. I’m Alex Kacik, senior operations reporter. I’m joined today by our technology reporter, Jessica Kim Cohen, to discuss the latest tech development in joint replacement surgeries. Thanks for joining me, Jessica.
Jessica Kim Cohen: Yeah, thanks for having me.
Alex Kacik: Jessica, you came out with a story recently about a “smart” knee implant. It’s the first of its kind, at least to be approved by the Food and Drug Administration. And it seems to be an embedded extension of remote monitoring. You know, we hear so much about internet connected things, especially in healthcare that are supposed to be making our lives easier, but let’s be real with me for a second. Are the robots taking over?
Alex Kacik: On that note, you talked to an analyst who said that the potential is significant, but it’ll take large research studies to prove the value to payers and providers. So let’s unpack that a bit.
Related Articles