How Does An Artificial Knee Replacement Work
The knee joint is formed where the femur meets the tibia and fibula . The patella, or kneecap, moves against the femur and rests atop the knee. Ligaments, muscles and cartilage bridge these bones and form a hinge, which creates flexibility and allows for movement.
When it comes to knee implants, only the tibia, patella and femur are affected, whether by resurfacing or replacing. Knee replacements are made in a variety of plastics, ceramics and metals:
Tibial component: This part is attached to the tibia with a stem inserted into the bone for stability. These pieces are generally in two parts and consist of a mix of plastic and metal.
Femoral component: This attaches to the end of the femur, allowing the patellar component to slide up and down when the knee bends.
Patellar component: This piece is intended to match the resurfaced patella and must properly align with the femoral component in order to work correctly. Tendons hold this piece in place.
Enjuris tip:
The knee replacement components will be inserted into place and sometimes cemented for additional stability. If the patient is younger, the doctor will likely opt out of using cement because the patients bone structure is strong enough without it.
Why People Are Filing Knee Replacement Lawsuits
Knee replacement lawsuits occur when knee implants fail or are defective. Patients may receive knee implants during knee replacement surgery, also known as total knee arthroplasty. If the implants are defective, patients may experience negative side effects or implant failure.
Depending on the severity of side effects, recipients of faulty knee implants may require costly medical care or additional surgeries. Knee implant lawsuits allow such patients to hold the manufacturer financially responsible for defective knee implants.
How Do Knee Replacements Help People
Knee replacements have improved the quality of life for millions of patients, however, some patients ultimately experience more harm than benefit. Like many other joint replacement procedures, knee replacements use medical devices that are frequently found to be defective resulting in complications that create severe adverse events and may require revision surgery.
Recommended Reading: Which Treadmill Is Best For Knees
What To Do If You Think You May Have A Knee Replacement Lawsuit
First, talk with your surgeon to see what must be done if you are experiencing pain in one or both of your knees. You might be able to fix the issue without surgical intervention. The most important thing is to get you out of pain and make sure your implant is not failing.
If youve already explored pain from your knee replacement implant with your doctor, talk to an attorney.
Each state has a statute of limitations, which is the time available to file your knee replacement lawsuit. Once you are past that time frame, you lose the ability to file. Some states only offer one year for cases, so get this done. A lawyer will be able to preserve your claim.
Personal Injury Attorney Interview SheetWorksheet with questions to ask a personal injury attorney to help determine if he or she will be a good fit for your case
Keep in mind, however, that defective products cases take a long, long time to finish. It might be a decade before you see any money, if at all. Make sure not to get your hopes too high, too soon.
Products liability law is unique. Youll want to look for a products liability/defective products lawyer who has many years of experience, preferably also with knee replacement lawsuits.
If you have not met with a lawyer yet, consider looking through the Enjuris lawyer directory for someone in your state who can help.
A Brief History Of Knee Replacement Implants
The total knee replacement is one of the most important surgical advances of the 20th century. Knee replacements, believe it or not, date back to the late 1800s.
In 1890, German surgeon Themistocles Gluck was the first to surgically implant joint hinges made of ivory.
The Walldius hinge joint was created in 1951, originally manufactured in acrylic before changing to cobalt and chrome in 1958.
This hinge joint suffered from early failure, so surgeons and developers kept trying. This led John Charnley to create the cemented metal-on-polyethylene total hip arthroplasty in the early 1960s. Improvements kept being made in the schematics of the device, where they were placed and what materials were used, which brings us to the modern-day knee replacement implants.
Read Also: Left Side Of Left Knee Pain
Smith & Nephew Knee Replacement Lawsuits
Some Smith & Nephew knee replacement systems have been linked to a high number of failures and other problems that could lead to the need for revision surgery.
SMITH & NEPHEW KNEE REPLACEMENT LAWSUIT STATUS: Potential claims are being reviewed by Smith & Nephew knee lawyers for individuals who have experienced problems after a knee replacement involving certain Smith & Nephew Journey knee components.
Complications Related To Pain After Knee Replacement Surgery
Most patients who receive a total or partial knee replacement end up re-joining life at a better pace, as theyre able to move without pain and walk more normally. However, that is not to say that complications and pain after knee replacement surgery dont occur. We all know that even the most routine medical procedures can cause problems.
Here are the most common issues that patients report. These may lead to pain after knee replacement surgery or other symptoms:
- Device loosening
- Metallosis : the metal implant pieces rub together, releasing particles into the bloodstream and joint
- Joint infection
You May Like: How To Get Fluid Off Knee
Study Links Journey Deuce To High Failure Rate
The Journey of Arthroplasty has published a study of 36 people implanted with the Smith & Nephew Journey Deuce knee implant. They found that 14% of patients needed revision surgery within two years, including one patient who had a catastrophically failed tibial baseplate. In addition, 31% of patients reported poor results, and 53% said they would not have the surgery again.
Smith & Nephew Journey I Bcs Knee Recall
In June 2018, Smith & Nephew voluntarily issued a recall for the Journey I BCS Knee. In the Urgent Field Safety Notice the company released, it explains that the knee was found to have a revision rate one and a half times higher than similar knee implants. The company further stated that recipients of the device could need revision surgery earlier than they or their doctors would normally expect. The Notice also cautioned medical practitioners and facilities that had purchased the unit to find and set aside any unused devices.
The U.S. Food and Drug Administration later classified this recall as a Class II recall: a situation in which use of or exposure to a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.
Don’t Miss: Popping Sound In Knee With Pain
Complaint Claims 288 Device Problems At Time Of Recall
In January 2011, Alberto Grazia received a Smith & Nephew hip implant at Euclid Hospital in Euclid, Ohio. A Birmingham Hip Resurfacing system replaced his right hip. Grazias left hip was also replaced with a BHR system in July 2011.
Grazia endured severe pain after his second hip implant. It was so bad that surgeons had to replace his left hip implant in April 2016.
In December 2016, Grazia filed a lawsuit against Smith & Nephew. It was later included in the MDL over BHR implants. He claimed metal shedding debris from friction between the hip-replacement parts led to his persistent pain.
The lawsuit claimed at the time of the devices recall in 2015, there had been 288 device problems with the BHR, including numerous safety problems related to metal shedding debris and other symptoms typical of metal-on-metal device failure.
What Are The Benefits Of Filing A Knee Replacement Lawsuit
When a patient is injured by a defective medical device, that patient is faced with the decision of whether or not to pursue a lawsuit. Our lawyers are here to help you make that decision and explain what your options are. If you have a viable case, our firm will represent you on a contingency fee basis meaning you dont pay anything until we obtain compensation for you. Anyone that files a knee replacement lawsuit may receive compensation for the following:
- Medical expenses
- Loss of income due to extended recovery times
- Loss of companionship with family members
- Corrective surgery to either repair or replace the existing elbow implant
- Pain and suffering
Don’t Miss: What To Do For Gout In Your Knee
New York City Injury Attorneys
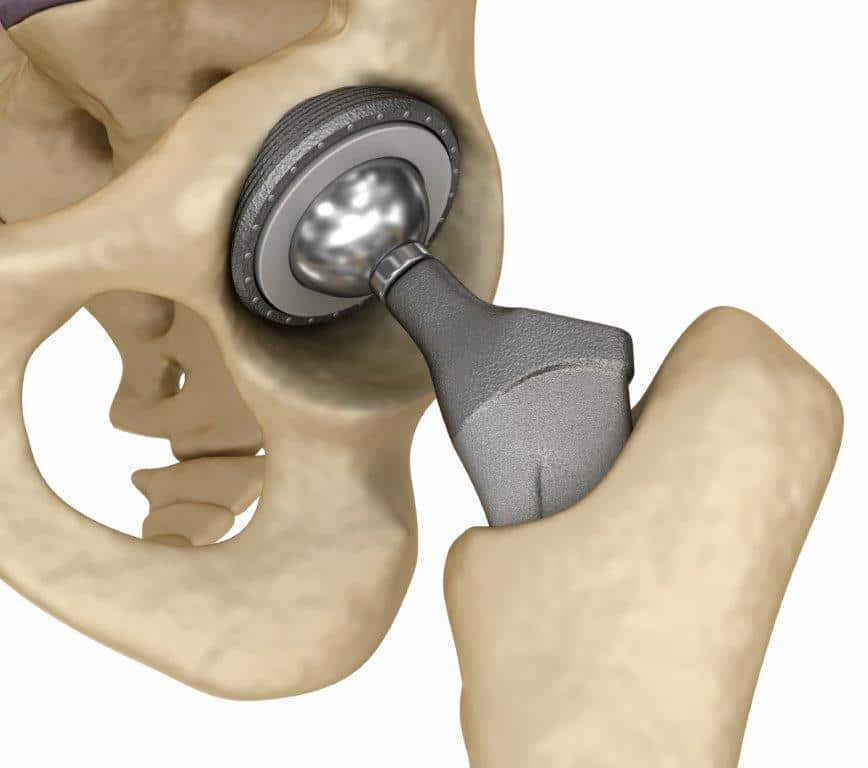
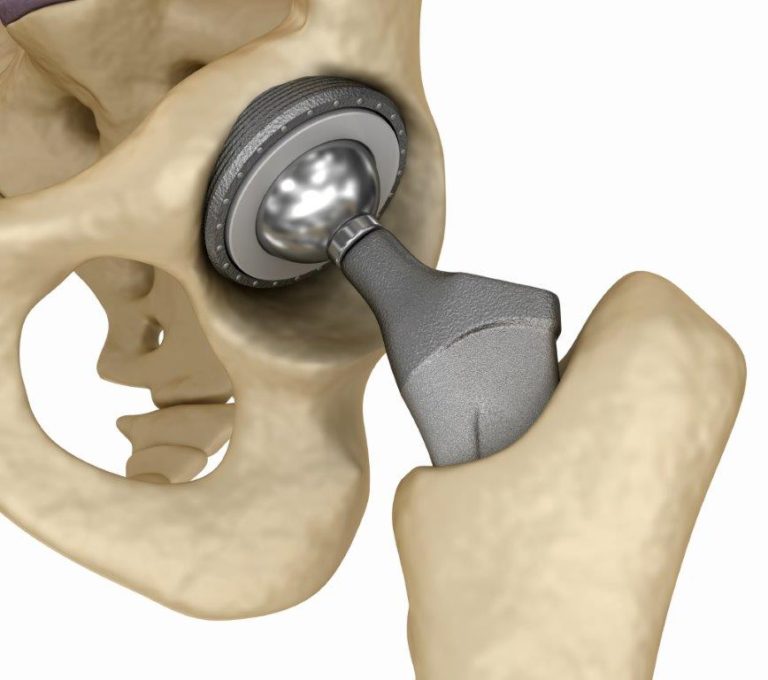
Smith & Nephew recalled the R3 hip replacement system, which consists of a metal ball implanted into the end of the femur that articulates with a metal cup on the pelvis. The company also markets the Birmingham Hip Replacement , a hip resurfacing system which involves a metal plating over the existing femur ball. The R3 has been recalled. The BHR has not been recalled, but it has generated similar complaints of premature failure, metal sensitivity and tissue damage when the BHR liner is coupled with the R3 system.
Smith & Nephew previously recalled its Oxinium knee replacement system because of widespread problems. You may have legal remedies if you were affected by the Smith & Nephew knee replacement recall.
Our firm also has an office in San Juan, Puerto Rico. today to schedule your consultation.
Thank you for contacting us. We will get back to you shortly.
Please complete all required fields below.
This Content Is For Informational Purposes Only
Trantolo & Trantolo is not currently accepting cases for this lawsuit. Please check in the future for any updates.
In recent years, manufacturers have introduced new knee replacement devices. These implants replace the cement bonding of the past a practice that potentially broke the attached bone with an artificial joint that fuses to the skeleton. The device features outputs for the femoral, tibia, and patella.
However, multiple recalls and studies have shed a negative light onto this medical development. A replacement joint, regardless of design, must support a patients body weight while allowing for mobility, and is built to provide 15 years of usage. In 2011, an Austrian study contradicted this latter statement, revealing that newer knee implants may be 30 percent less strong and have higher-than-average revision rates.
As a result, patients given these newer implants have a greater chance of experiencing a loose joint, severe pain, infection, inflammation, swelling, and neurological damage.
So far, the FDA has recalled 118 knee replacements or components over damaged parts, disassociation, mislabeled components, seize, early wear, and delamination. Some of these include:
- Biomet Vanguard CR
- Wear and tear on the joint
In response, patients have needed revisions surgery, a procedure that may lead to additional complications: infection, thrombophlebitis, myositis ossificans, bone fractions, dislocation, a difference in leg length, and faster loss of bone tissue.
Don’t Miss: When Can I Drive After Knee Replacement
Recalls For Knee Replacement Implants
There have been many knee implant devices recalled over the years. Reasons range from device loosening to packaging defects, the latter of which might be a reason you never heard about a specific recall. The recall could have been initiated by the Food & Drug Administration, but its far more common that a company recalled its own product for a specific reason.
Enjuris tip:
The following companies have had numerous instances when they needed to claw back a product from public commerce:
- Biomet: 75 recalls
Lawsuit Filed Over Smith & Nephew Total Knee Replacement Failure
A Louisiana man has filed a lawsuit over a Smith & Nephew total knee replacement, which allegedly failed less than one year after surgery.
The complaint was filed by Henry Bias against Smith & Nephew, Inc. in the U.S. District Court for the Western District of Louisiana on March 29.
According to the product liability lawsuit, Bias received a Smith & Nephew Genesis II knee replacement system on September 24, 2009, and by April 29, 2010 he was required to undergo revision surgery due to failure of the implants components. He then had to undergo surgery again on March 31, 2011, as a result of problems with the total knee replacement.
Also Check: Can You Get Shingles On Your Knee
Smith & Nephew Knee Replacement Mass Tort Lawyers
Potential Smith & Nephew knee lawsuits are being investigated and reviewed for individuals throughout the United States. All cases are pursued by our law firm under a contingency fee agreement, which means that there are never any fees unless a recovery is obtained.
It appears that Smith & Nephew may have failed to take adequate steps to properly research their product, reduce the risk of injury or warn consumers. As a result, financial compensation may be available for Smith & Nephew Journey knee complications that were caused by a broken tibial base plate or other defective components.
To review a potential claim for yourself, a friend or family member, request a free consultation and claim evaluation.
Get My Free Consultation
Complications Mentioned In Knee Device Lawsuits
- Pain and aching
Due to the volume of lawsuits, many of these cases were consolidated into multidistrict litigation. Plaintiffs seek legal damages related to medical bills, pain and suffering related to their faulty knee implants.
Multidistrict litigation is another method that courts use to merge lawsuits. Unlike a class action lawsuit, individual cases remain separate in an MDL. However, certain phases of litigation are combined for all the lawsuits. This speeds up the process of trying hundreds of similar cases.
Recommended Reading: What To Know About Knee Replacement
Have You Had Complications With R3 Hip Implant Or Bhr Birmingham
Smith & Nephew, one of the major players in orthopedic implants, announced in June 2012 that it was recalling its popular hip replacement systems, including hip joints already implanted in patients throughout the world. The Smith & Nephew recall is the latest, and probably not the last, by manufacturers of metal-on-metal hip implants.
Touted as more durable than ceramic hip replacements, metal-on-metal hip replacements as a class have had a high rate of revision surgery and a high incidence of metallosis . At Sullivan & Brill, LLP, our personal injury lawyers can handle your possible claim against Smith & Nephew if you have suffered complications of your R3 or BHR hip replacement, including the need for revision surgery. We represent people in New York City, statewide New York, and nationwide. Contact us today for a free consultation.
What Serious Knee Replacement Complications Can Occur
More severe complications from knee replacement may include serious and possibly life-threatening effects such as:
- Persistent or increasing knee pain
- Infection with fever or chills
- Blood clots creating pain in the lower leg
- Nerve and blood vessel damage which may be permanent
- Knee replacement failure resulting in dislocation and severe pain
- Periprosthetic fractures in the bone surrounding the implant
- Particle shedding from device structure that is deposited into surrounding tissue
- Osteolysis or necrosis caused by shed particles
How Can a Knee Replacement Cause an Infection?
Knee replacement infections can range from minor to severe. Typically, medical professionals sterilize the knee before the operation and use sterile equipment and practices during the procedure. However, the open wound can still trap bacteria. Patients with a knee replacement infection may experience fever. Tenderness, redness, and swelling around the infection site are also common.
How Can a Knee Replacement Cause Blood Clots?
Blood clots are a potential knee replacement complication. Blood clots occur when blood cells and other substances form clumps that can block the patients blood vessels. During knee replacement surgery, the blood around the knee may become stagnant or move slowly. This is a common cause of knee replacement complications such as blood clots. Blood thinners or special stockings may help the patients blood move faster. These treatments can be used during and after surgery.
You May Like: Crunching Sound In Knee With Pain Treatment
How Weitz & Luxenberg Can Help
Weitz & Luxenberg is currently accepting clients who received a first generation Smith & Nephew Journey BCS knee and subsequently needed to undergo a revision, or replacement, surgery or have been advised by their doctor they need to undergo a revision surgery due to femoral component loosening. We invite you to contact us for more information about your legal options.
If you have undergone a knee replacement surgery, are having problems, and are not sure whether you received a first generation Smith & Nephew Journey BCS knee, we encourage you to ask your doctor which knee replacement model you received. Depending on your specific implant, you may be able to seek compensation.
When Are Knee Replacements Needed

Knee replacements are required due to an injured joint or one that has simply worn out. Conditions such as arthritis and developmental abnormalities may cause the need for a knee replacement, but injuries and acute medical conditions also contribute. Knee replacement surgeries are recommended when pain and lack of mobility interfere with normal daily activities.
Read Also: How Long After Knee Surgery Can You Drive
What Is A Mobile Bearing Knee Replacement
A mobile-bearing knee replacement can use either a rotating platform or a medial pivot. Mobile bearing and rotating platform implants provide some flexibility by allowing the tibial component plastic cushion to rotate. This gives greater flexibility on the sides of the knee and is used in young and active patients. Mobile bearing, medial pivot rotates and flexes like the natural knee joint and is also used for younger, more active patients.
What Zimmer Biomet Knee Replacement Lawsuits Are There
Zimmer currently faces more than 700 lawsuits for its NexGen knee replacement system. Federal cases filed against Zimmer were consolidated into multidistrict litigation . An MDL is used to streamline the pretrial process for a number of similar cases. The Zimmer MDL was transferred to the U.S. District Courts Northern District of Illinois. After the pretrial phase is complete, each case will be returned to its respective district.
Don’t Miss: What Are The Different Types Of Knee Injections